声明不存在利益冲突参考文献
[1] Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the global burden of disease study 2017[J]. Lancet, 2018, 392(10159):1736-1788. DOI: 10.1016/S0140-6736(18)32203-7.
[2] Pilote L, Beck CA, Karp I, et al. Secondary prevention after acute myocardial infarction in four Canadian provinces, 1997-2000[J]. Can J Cardiol, 2004, 20(1):61-67.
[3] Al-Omran M, Mamdani MM, Lindsay TF, et al. Suboptimal use of statin therapy in elderly patients with atherosclerosis: a population-based study[J]. J Vasc Surg, 2008, 48(3):607-612. DOI: 10.1016/j.jvs.2008.04.057.
[4] He J, Gu D, Reynolds K, et al. Serum total and lipoprotein cholesterol levels and awareness, treatment, and control of hypercholesterolemia in China[J]. Circulation, 2004, 110(4):405-411. DOI: 10.1161/01.CIR.0000136583.52681.0D.
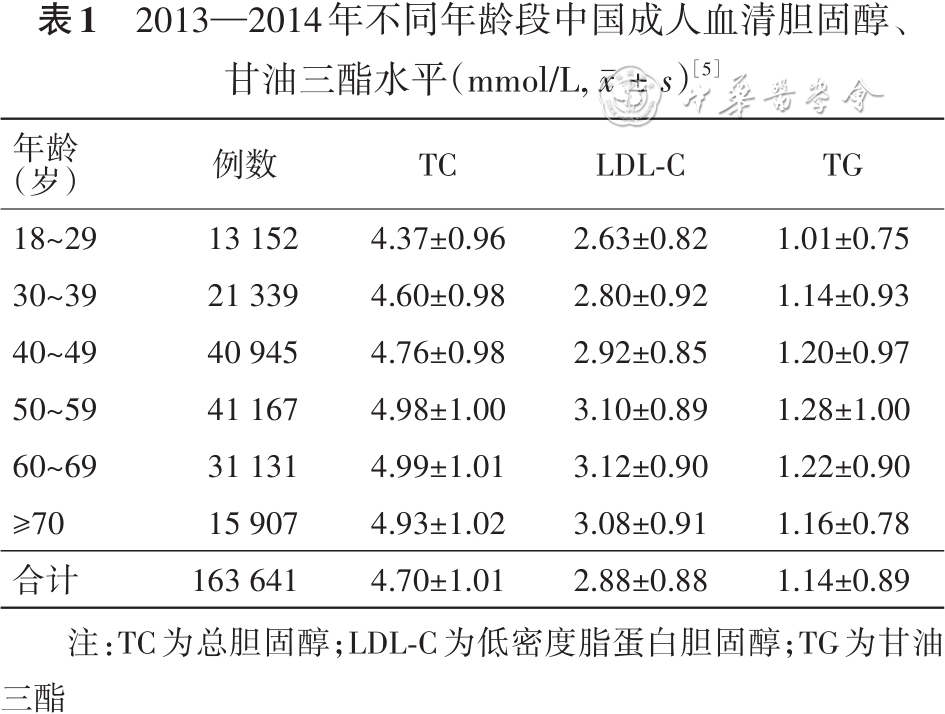
[5] Zhang M, Deng Q, Wang L, et al. Prevalence of dyslipidemia and achievement of low-density lipoprotein cholesterol targets in Chinese adults: a nationally representative survey of 163, 641 adults[J]. Int J Cardiol, 2018, 260:196-203. DOI: 10.1016/j.ijcard.2017.12.069.
[6] Yusuf S, Bosch J, Dagenais G, et al. Cholesterol lowering in intermediate-risk persons without cardiovascular disease[J]. N Engl J Med, 2016, 374(21):2021-2031. DOI: 10.1056/NEJMoa1600176.
[7] Cholesterol Treatment Trialists′ Collaboration. Efficacy and safety of statin therapy in older people: a meta-analysis of individual participant data from 28 randomised controlled trials[J]. Lancet, 2019, 393(10170):407-415. DOI: 10.1016/S0140-6736(18)31942-1.
[8] Mortensen MB, Nordestgaard BG. Elevated LDL cholesterol and increased risk of myocardial infarction and atherosclerotic cardiovascular disease in individuals aged 70-100 years: a contemporary primary prevention cohort[J]. Lancet, 2020, 396(10263):1644-1652. DOI: 10.1016/S0140-6736(20)32233-9.
[9] Orkaby AR, Driver JA, Ho YL, et al. Association of statin use with all-cause and cardiovascular mortality in US veterans 75 years and older[J]. JAMA, 2020, 324(1):68-78. DOI: 10.1001/jama.2020.7848.
[10] Gencer B, Marston NA, Im K, et al. Efficacy and safety of lowering LDL cholesterol in older patients: a systematic review and meta-analysis of randomised controlled trials[J]. Lancet, 2020, 396(10263):1637-1643. DOI: 10.1016/S0140-6736(20)32332-1.
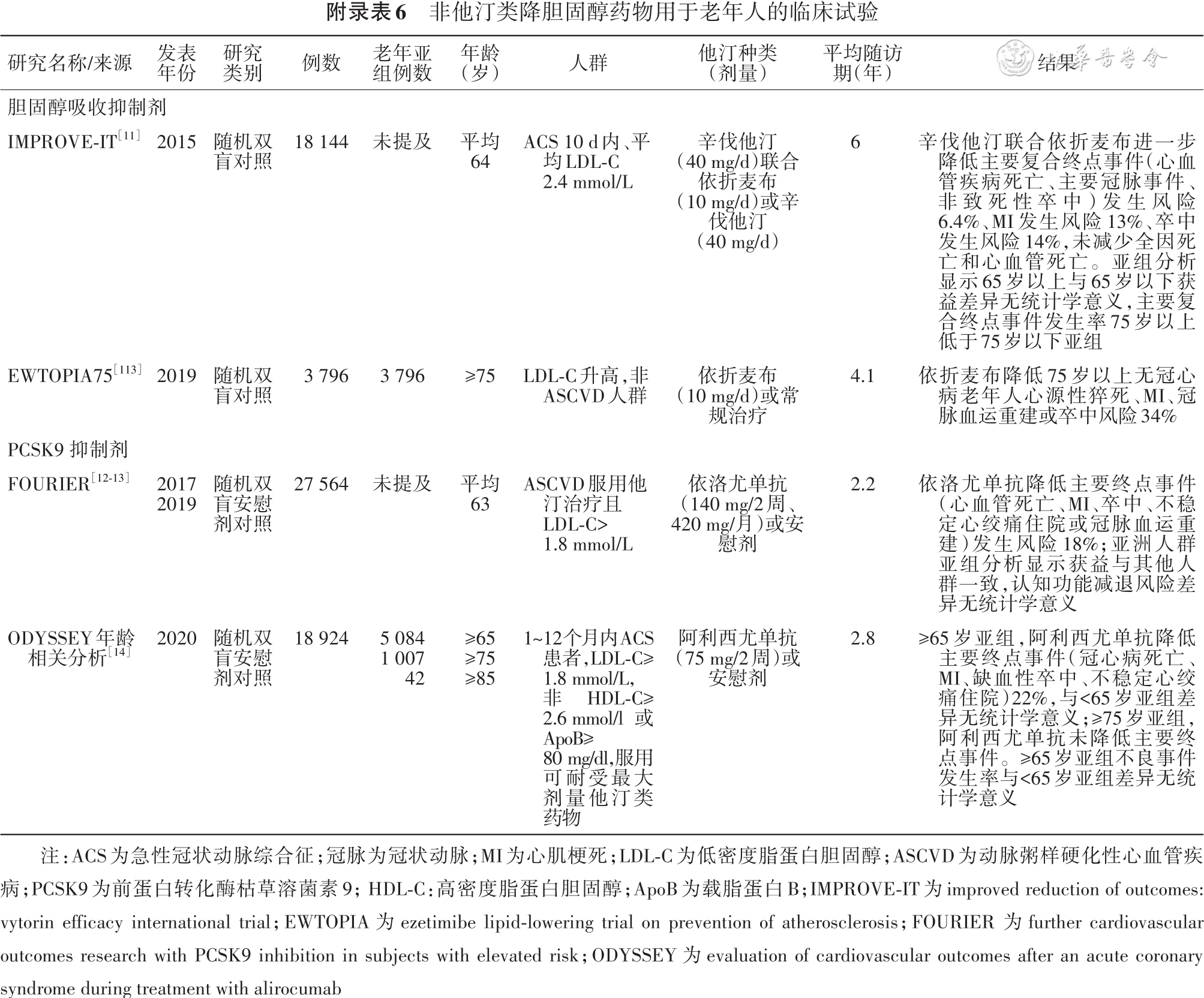
[11] Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes[J]. N Engl J Med, 2015, 372(25):2387-2397. DOI: 10.1056/NEJMoa1410489.
[12] Murphy SA, Pedersen TR, Gaciong ZA, et al. Effect of the PCSK9 inhibitor evolocumab on total cardiovascular events in patients with cardiovascular disease: a prespecified analysis from the FOURIER trial[J]. JAMA Cardiol, 2019, 4(7):613-619. DOI: 10.1001/jamacardio.2019.0886.
[13] Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease[J]. N Engl J Med, 2017, 376(18):1713-1722. DOI: 10.1056/NEJMoa1615664.
[14] Sinnaeve PR, Schwartz GG, Wojdyla DM, et al. Effect of alirocumab on cardiovascular outcomes after acute coronary syndromes according to age: an ODYSSEY OUTCOMES trial analysis[J]. Eur Heart J, 2020, 41(24):2248-2258. DOI: 10.1093/eurheartj/ehz809.
[15] Hokanson JE, Austin MA. Plasma triglyceride level is a risk factor for cardiovascular disease independent of high-density lipoprotein cholesterol level: a meta-analysis of population-based prospective studies[J]. J Cardiovasc Risk, 1996, 3(2): 213-219.
[16] Iso H, Imano H, Yamagishi K, et al. Fasting and non-fasting triglycerides and risk of ischemic cardiovascular disease in Japanese men and women: the Circulatory Risk in Communities Study (CIRCS)[J]. Atherosclerosis, 2014, 237(1):361-368. DOI: 10.1016/j.atherosclerosis.2014.08.028.
[17] Kim EH, Lee JB, Kim SH, et al. Serum triglyceride levels and cardiovascular disease events in Koreans[J]. Cardiology, 2015, 131(4):228-235. DOI: 10.1159/000380941.
[18] Reiner ?. Hypertriglyceridaemia and risk of coronary artery disease[J]. Nat Rev Cardiol, 2017, 14(7):401-411. DOI: 10.1038/nrcardio.2017.31.
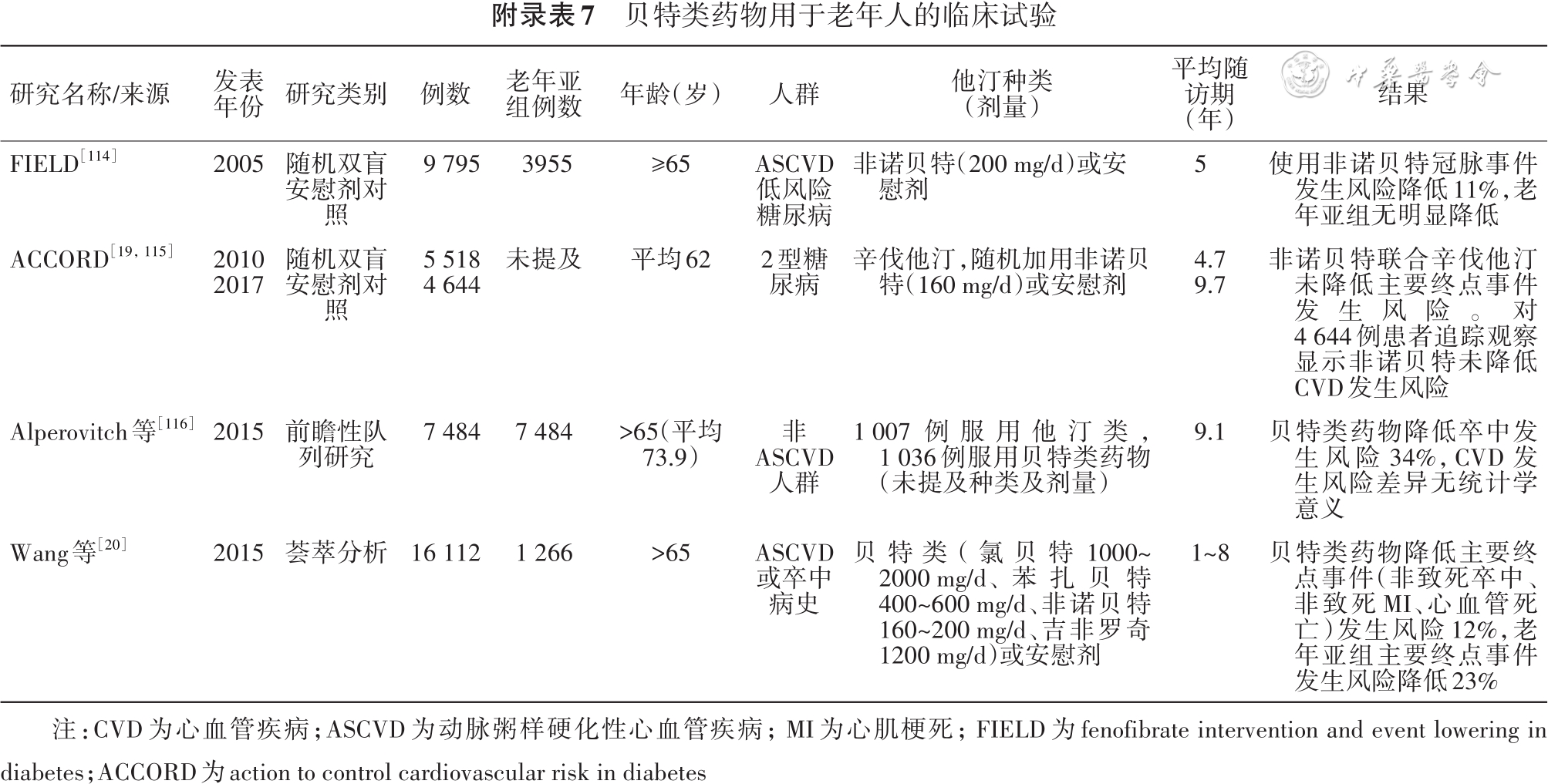
[19] Ginsberg HN, Elam MB, Lovato LC, et al. Effects of combination lipid therapy in type 2 diabetes mellitus[J]. N Engl J Med, 2010, 362(17):1563-1574. DOI: 10.1056/NEJMoa1001282.
[20] Wang D, Liu B, Tao W, et al. Fibrates for secondary prevention of cardiovascular disease and stroke[J]. Cochrane Database Syst Rev, 2015, 2015(10):CD009580. DOI: 10.1002/14651858.CD009580.pub2.
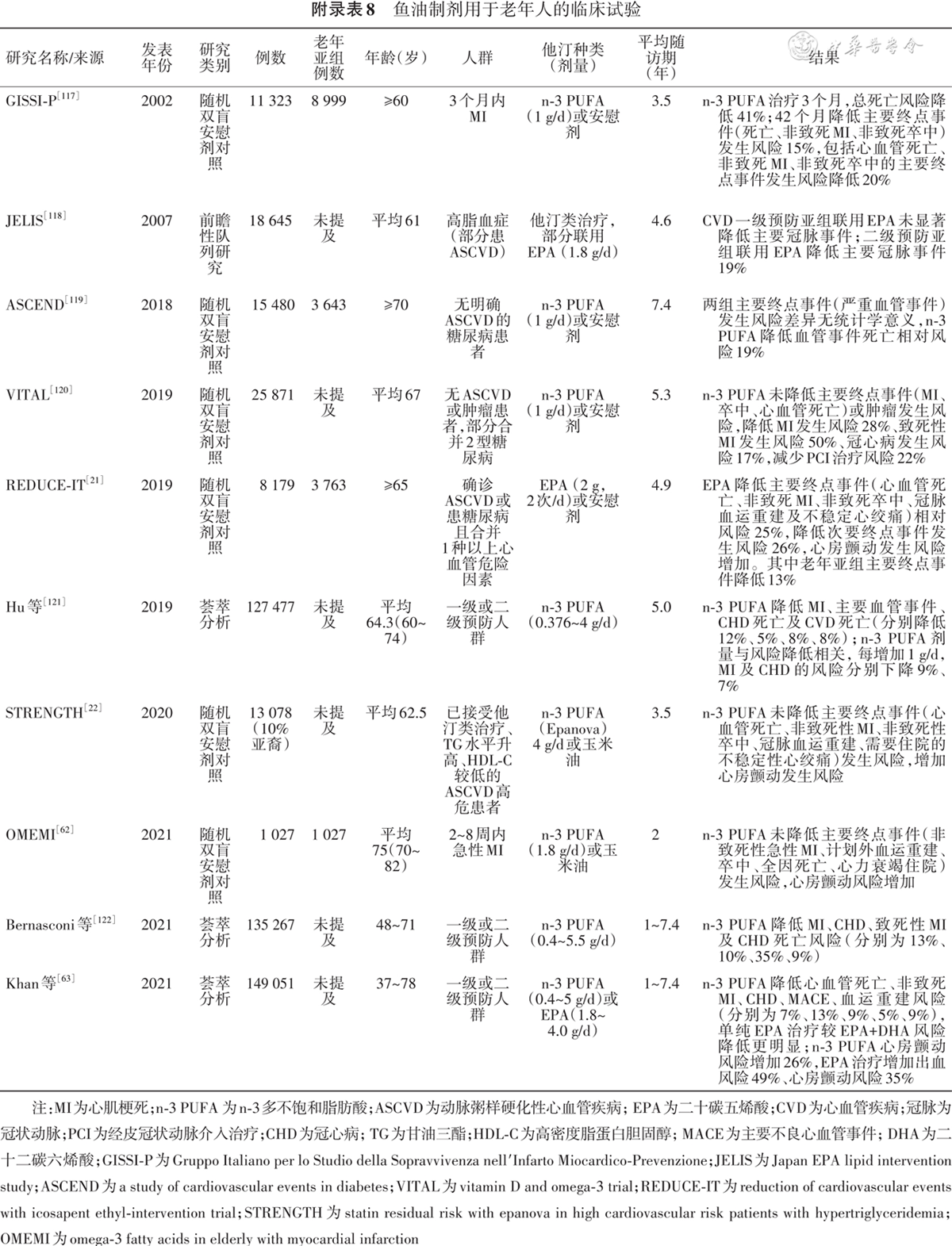
[21] Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia[J]. N Engl J Med, 2019, 380(1):11-22. DOI: 10.1056/NEJMoa1812792.
[22] Nicholls SJ, Lincoff AM, Garcia M, et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: the STRENGTH randomized clinical trial[J]. JAMA, 2020, 324(22):2268-2280. DOI: 10.1001/jama.2020.22258.
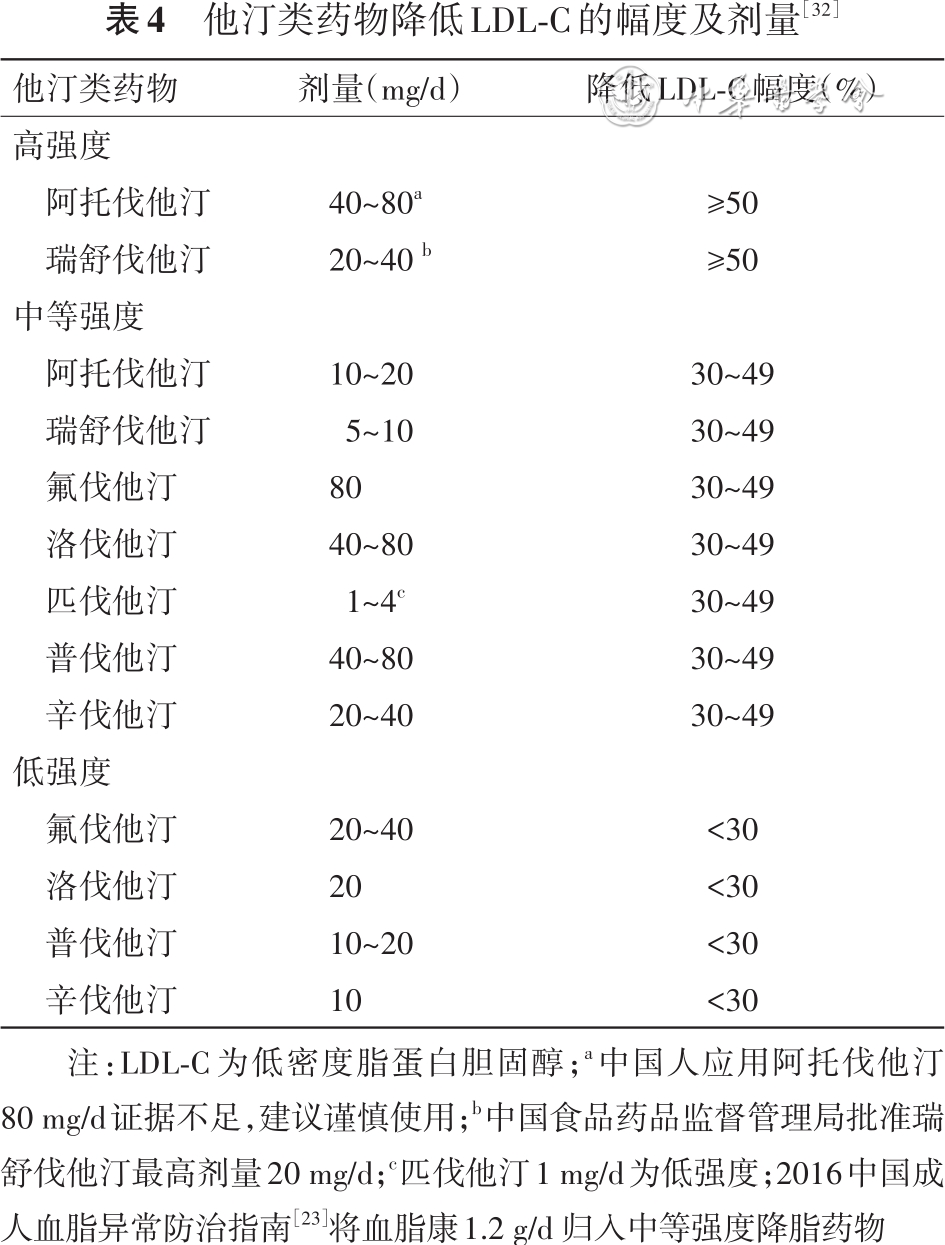
[23] 中国成人血脂异常防治指南修订联合委员会. 中国成人血脂异常防治指南(2016年修订版)[J]. 中华心血管病杂志, 2016, 44(10):833-853. DOI: 10.3760/cma.j.issn.0253-3758.2016.10.005.
[24] Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines[J]. J Am Coll Cardiol, 2019, 73(24): e285-e350. DOI: 10.1016/j.jacc.2018.11.003.
[25] Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines[J]. Circulation, 2019, 140(11):e596-e646. DOI: 10.1161/CIR.0000000000000678.
[26] Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk[J]. Eur Heart J, 2020, 41(1): 111-188. DOI: 10.1093/eurheartj/ehz455.
[27] Visseren F, Mach F, Smulders YM, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice[J]. Eur Heart J, 2021, 42(34):3227-3337. DOI: 10.1093/eurheartj/ehab484.
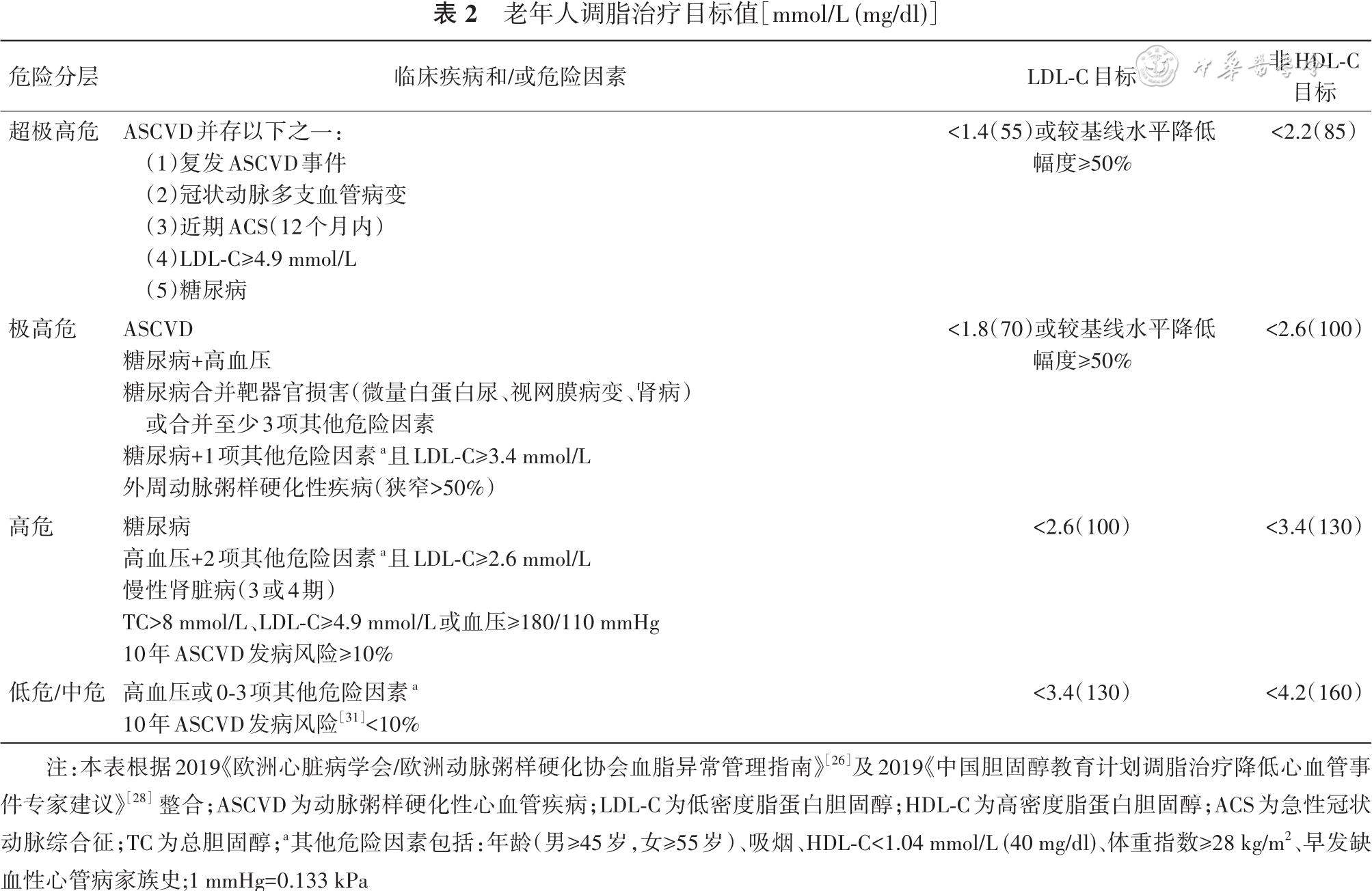
[28] 中国胆固醇教育计划(CCEP)工作委员会, 中国医疗保健国际交流促进会动脉粥样硬化血栓疾病防治分会, 中国老年学和老年医学学会心血管病分会, 等. 中国胆固醇教育计划调脂治疗降低心血管事件专家建议(2019)[J]. 中华内科杂志, 2020, 59(1):18-22. DOI: 10.3760/cma.j.issn.0578-1426.2020.01.016.
[29] 中华医学会心血管病学分会动脉粥样硬化与冠心病学组, 中华心血管病杂志编辑委员会. 超高危动脉粥样硬化性心血管疾病患者血脂管理中国专家共识[J]. 中华心血管病杂志, 2020, 48(4):280-286. DOI: 10.3760/cma.j.cn112148-20200121-00036.
[30] Virani SS, Morris PB, Agarwala A, et al. 2021 ACC Expert consensus decision pathway on the management of ASCVD risk reduction in patients with persistent hypertriglyceridemia: a report of the American College of Cardiology Solution Set Oversight Committee[J]. J Am Coll Cardiol, 2021, 78(9):960-993. DOI: 10.1016/j.jacc.2021.06.011.
[31] 王淼, 刘静, 赵冬. 中国动脉粥样硬化性心血管病发病危险评估的新方案[J]. 中华心血管病杂志, 2018, 46(2):87-91. DOI: 10.3760/cma.j.issn.0253-3758.2018.02.003.
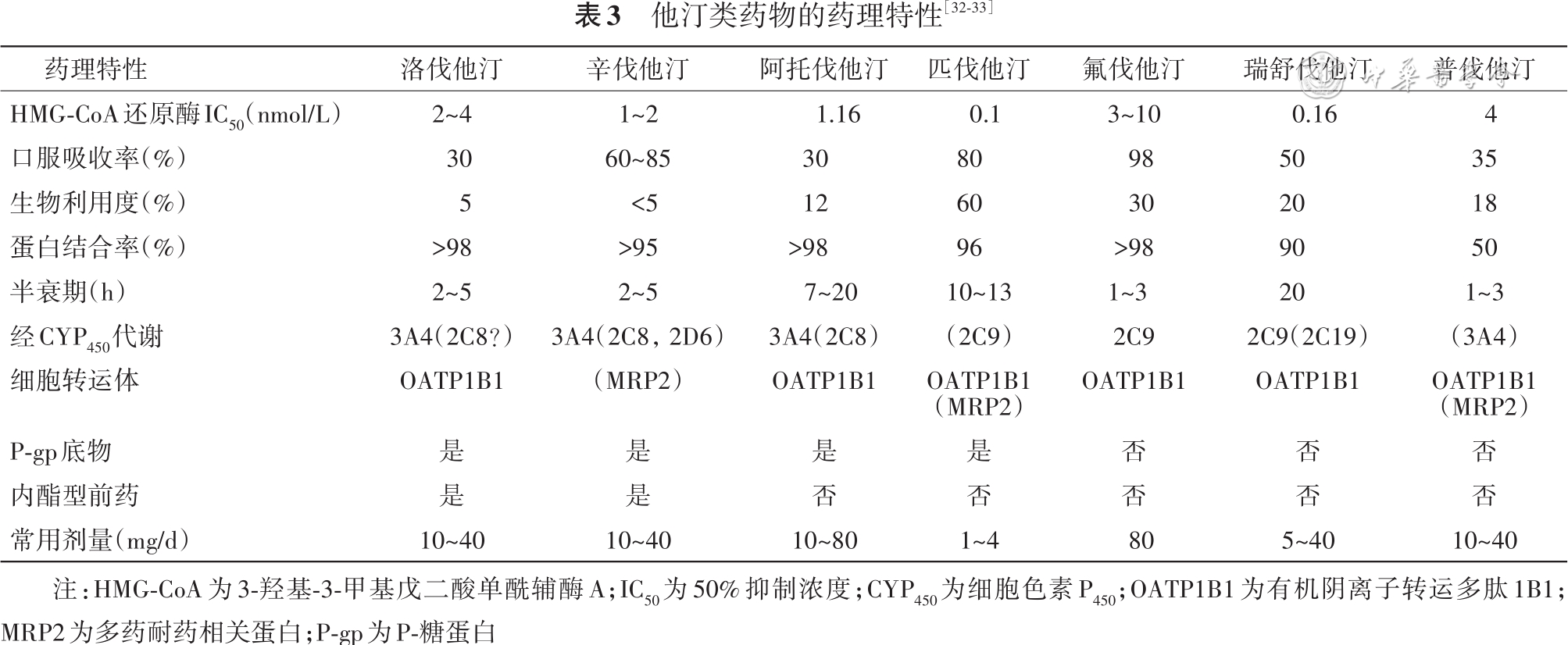
[32] Newman CB, Preiss D, Tobert JA, et al. Statin safety and associated adverse events: a scientific statement from the American Heart Association[J]. Arterioscler Thromb Vasc Biol, 2019, 39(2):e38-e81. DOI: 10.1161/ATV.0000000000000073.
[33] Mach F, Ray KK, Wiklund O, et al. Adverse effects of statin therapy: perception vs. the evidence-focus on glucose homeostasis, cognitive, renal and hepatic function, haemorrhagic stroke and cataract[J]. Eur Heart J, 2018, 39(27): 2526-2539. DOI: 10.1093/eurheartj/ehy182.
[34] Cai T, Abel L, Langford O, et al. Associations between statins and adverse events in primary prevention of cardiovascular disease: systematic review with pairwise, network, and dose-response meta-analyses[J]. BMJ, 2021, 374: n1537. DOI: 10.1136/bmj.n1537.
[35] Kellick KA, Bottorff M, Toth PP, et al. A clinician′s guide to statin drug-drug interactions[J]. J Clin Lipidol, 2014, 8(3 Suppl): S30-S46. DOI: 10.1016/j.jacl.2014.02.010.
[36] Liu J, Liang W, Jing W, et al. Countdown to 2030: eliminating hepatitis B disease, China[J]. Bull World Health Organ, 2019, 97(3):230-238. DOI: 10.2471/BLT.18.219469.
[37] 他汀类药物安全性评价工作组. 他汀类药物安全性评价专家共识[J]. 中华心血管病杂志, 2014, 42(11): 890-894. DOI: 10.3760/cma.j.issn.0253-3758.2014.11.002.
[38] Schech S, Graham D, Staffa J, et al. Risk factors for statin-associated rhabdomyolysis[J]. Pharmacoepidemiol Drug Saf, 2007, 16(3): 352-358. DOI: 10.1002/pds.1287.
[39] Agarwal R. Effects of statins on renal function[J]. Am J Cardiol, 2006, 97(5):748-755. DOI: 10.1016/j.amjcard.2005.09.110.
[40] Sidaway JE, Davidson RG, McTaggart F, et al. Inhibitors of 3-hydroxy-3-methylglutaryl-CoA reductase reduce receptor-mediated endocytosis in opossum kidney cells[J]. J Am Soc Nephrol, 2004, 15(9):2258-2265. DOI: 10.1097/01.ASN.0000138236.82706.EE.
[41] Waters DD, Ho JE, Boekholdt SM, et al. Cardiovascular event reduction versus new-onset diabetes during atorvastatin therapy: effect of baseline risk factors for diabetes[J]. J Am Coll Cardiol, 2013, 61(2):148-152. DOI: 10.1016/j.jacc.2012.09.042.
[42] Cho L, Rocco M, Colquhoun D, et al. Clinical profile of statin intolerance in the phase 3 GAUSS-2 study[J]. Cardiovasc Drugs Ther, 2016, 30(3):297-304. DOI: 10.1007/s10557-016-6655-4.
[43] 血脂异常老年人使用他汀类药物中国专家共识组, 刘梅林, 胡大一. 血脂异常老年人使用他汀类药物中国专家共识 [J]. 中华内科杂志, 2015, 54(5): 467-477. DOI: 10.3760/cma.j.issn.0578-1426.2015.05.020.
[44] Vallianou NG, Kostantinou A, Kougias M, et al. Statins and cancer[J]. Anticancer Agents Med Chem, 2014, 14(5):706-712. DOI: 10.2174/1871520613666131129105035.
[45] Phan BA, Dayspring TD, Toth PP. Ezetimibe therapy: mechanism of action and clinical update[J]. Vasc Health Risk Manag, 2012, 8:415-427. DOI: 10.2147/VHRM.S33664.
[46] Cicero AF, Tartagni E, Ertek S. Safety and tolerability of injectable lipid-lowering drugs: a review of available clinical data[J]. Expert Opin Drug Saf, 2014, 13(8):1023-1030. DOI: 10.1517/14740338.2014.932348.
[47] Lam JK, Chow MY, Zhang Y, et al. siRNA versus miRNA as therapeutics for gene silencing[J]. Mol Ther Nucleic Acids, 2015, 4(9): e252. DOI: 10.1038/mtna.2015.23.
[48] Fitzgerald K, White S, Borodovsky A, et al. A Highly durable RNAi therapeutic inhibitor of PCSK9[J]. N Engl J Med, 2017, 376(1): 41-51. DOI: 10.1056/NEJMoa1609243.
[49] Simha V. Management of hypertriglyceridemia[J]. BMJ, 2020, 371: m3109. DOI: 10.1136/bmj.m3109.
[50] Jun M, Zhu B, Tonelli M, et al. Effects of fibrates in kidney disease: a systematic review and meta-analysis[J]. J Am Coll Cardiol, 2012, 60(20):2061-2071. DOI: 10.1016/j.jacc.2012.07.049.
[51] Brunzell JD. Clinical practice. Hypertriglyceridemia[J]. N Engl J Med, 2007, 357(10):1009-1017. DOI: 10.1056/NEJMcp070061.
[52] Landray MJ, Haynes R, Hopewell JC, et al. Effects of extended-release niacin with laropiprant in high-risk patients[J]. N Engl J Med, 2014, 371(3):203-212. DOI: 10.1056/NEJMoa1300955.
[53] Boden WE, Probstfield JL, Anderson T, et al. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy[J]. N Engl J Med, 2011, 365(24):2255-2267. DOI: 10.1056/NEJMoa1107579.
[54] Sandesara PB, Virani SS, Fazio S, et al. The forgotten lipids: triglycerides, remnant cholesterol, and atherosclerotic cardiovascular disease risk[J]. Endocr Rev, 2019, 40(2): 537-557. DOI: 10.1210/er.2018-00184.
[55] Bays HE, Ballantyne CM, Kastelein JJ, et al. Eicosapentaenoic acid ethyl ester (AMR101) therapy in patients with very high triglyceride levels (from the multi-center, placebo-controlled, randomized, double-blind, 12-week study with an open-label extension [MARINE] trial)[J]. Am J Cardiol, 2011, 108(5):682-690. DOI: 10.1016/j.amjcard.2011.04.015.
[56] Harris WS, Ginsberg HN, Arunakul N, et al. Safety and efficacy of Omacor in severe hypertriglyceridemia[J]. J Cardiovasc Risk, 1997, 4(5-6): 385-391.
[57] Pownall HJ, Brauchi D, Kilin? C, et al. Correlation of serum triglyceride and its reduction by omega-3 fatty acids with lipid transfer activity and the neutral lipid compositions of high-density and low-density lipoproteins[J]. Atherosclerosis, 1999, 143(2):285-297. DOI: 10.1016/s0021-9150(98)00301-3.
[58] Maki KC, McKenney JM, Reeves MS, et al. Effects of adding prescription omega-3 acid ethyl esters to simvastatin (20 mg/day) on lipids and lipoprotein particles in men and women with mixed dyslipidemia[J]. Am J Cardiol, 2008, 102(4):429-433. DOI: 10.1016/j.amjcard.2008.03.078.
[59] Mackness MI, Bhatnagar D, Durrington PN, et al. Effects of a new fish oil concentrate on plasma lipids and lipoproteins in patients with hypertriglyceridaemia[J]. Eur J Clin Nutr, 1994, 48(12): 859-865.
[60] Abdelhamid AS, Brown TJ, Brainard JS, et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease[J]. Cochrane Database Syst Rev, 2018, 11(11):CD003177. DOI: 10.1002/14651858.CD003177.pub4.
[61] Weinberg RL, Brook RD, Rubenfire M, et al. Cardiovascular impact of nutritional supplementation with omega-3 fatty acids: JACC focus seminar[J]. J Am Coll Cardiol, 2021, 77(5):593-608. DOI: 10.1016/j.jacc.2020.11.060.
[62] Kalstad AA, Myhre PL, Laake K, et al. Effects of n-3 fatty acid supplements in elderly patients after myocardial infarction: a randomized, controlled trial[J]. Circulation, 2021, 143(6):528-539. DOI: 10.1161/CIRCULATIONAHA.120.052209.
[63] Khan SU, Lone AN, Khan MS, et al. Effect of omega-3 fatty acids on cardiovascular outcomes: a systematic review and meta-analysis[J]. EClinicalMedicine, 2021, 38:100997. DOI: 10.1016/j.eclinm.2021.100997.
[64] Wiggins BS, Saseen JJ, Page RL, et al. Recommendations for management of clinically significant drug-drug interactions with statins and select agents used in patients with cardiovascular disease: a scientific statement from the American Heart Association[J]. Circulation, 2016, 134(21):e468-e495. DOI: 10.1161/CIR.0000000000000456.
[65] Tomlinson B, Chan P, Zhang Y, et al. Efficacy and safety of add on therapies in patients with hypercholesterolemia undergoing statin therapy[J]. Expert Opin Pharmacother, 2020, 21(17):2137-2151. DOI: 10.1080/14656566.2020.1801638.
[66] Feldman T, Davidson M, Shah A, et al. Comparison of the lipid-modifying efficacy and safety profiles of ezetimibe coadministered with simvastatin in older versus younger patients with primary hypercholesterolemia: a post Hoc analysis of subpopulations from three pooled clinical trials[J]. Clin Ther, 2006, 28(6):849-859. DOI: 10.1016/j.clinthera.2006.06.001.
[67] Giral P, Neumann A, Weill A, et al. Cardiovascular effect of discontinuing statins for primary prevention at the age of 75 years: a nationwide population-based cohort study in France[J]. Eur Heart J, 2019, 40(43):3516-3525. DOI: 10.1093/eurheartj/ehz458.
[68] Downs JR, Clearfield M, Weis S, et al. Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas coronary atherosclerosis prevention study[J]. JAMA, 1998, 279(20):1615-1622. DOI: 10.1001/jama.279.20.1615.
[69] Sever PS, Dahlof B, Poulter N R, et al. Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than-average cholesterol concentrations, in the Anglo-Scandinavian cardiac outcomes trial--lipid lowering arm (ASCOT-LLA): a multicentre randomised controlled trial[J]. Lancet, 2003, 361(9364):1149-1158. DOI: 10.1016/S0140-6736(03)12948-0.
[70] Glynn RJ, Koenig W, Nordestgaard BG, et al. Rosuvastatin for primary prevention in older persons with elevated C-reactive protein and low to average low-density lipoprotein cholesterol levels: exploratory analysis of a randomized trial[J]. Ann Intern Med, 2010, 152(8):488-496, W174. DOI: 10.7326/0003-4819-152-8-201004200-00005.
[71] Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein[J]. N Engl J Med, 2008, 359(21):2195-2207. DOI: 10.1056/NEJMoa0807646.
[72] Ford I, Murray H, Mccowan C, et al. Long-term safety and efficacy of lowering low-density lipoprotein cholesterol with statin therapy: 20-year follow-up of West of Scotland coronary prevention study[J]. Circulation, 2016, 133(11):1073-1080. DOI: 10.1161/CIRCULATIONAHA.115.019014.
[73] Lemaitre RN, Psaty BM, Heckbert SR, et al. Therapy with hydroxymethylglutaryl coenzyme a reductase inhibitors (statins) and associated risk of incident cardiovascular events in older adults: evidence from the cardiovascular health study[J]. Arch Intern Med, 2002, 162(12):1395-1400. DOI: 10.1001/archinte.162.12.1395.
[74] Orkaby AR, Gaziano J M, Djousse L, et al. Statins for primary prevention of cardiovascular events and mortality in older men[J]. J Am Geriatr Soc, 2017, 65(11):2362-2368. DOI: 10.1111/jgs.14993.
[75] Ramos R, Comas-Cufi M, Marti-Lluch R, et al. Statins for primary prevention of cardiovascular events and mortality in old and very old adults with and without type 2 diabetes: retrospective cohort study[J]. BMJ, 2018, 362: k3359. DOI: 10.1136/bmj.k3359.
[76] Eilat-Tsanani S, Mor E, Schonmann Y. Statin use over 65 years of age and all-cause mortality: a 10-year follow-up of 19 518 people[J]. J Am Geriatr Soc, 2019, 67(10):2038-2044. DOI: 10.1111/jgs.16060.
[77] Kim K, Lee CJ, Shim C, et al. Statin and clinical outcomes of primary prevention in individuals aged>75?years: the SCOPE-75 study[J]. Atherosclerosis, 2019, 284:31-36. DOI: 10.1016/j.atherosclerosis.2019.02.026.
[78] Zhou Z, Ofori-Asenso R, Curtis AJ, et al. Association of statin use with disability-free survival and cardiovascular disease among healthy older adults[J]. J Am Coll Cardiol, 2020, 76(1): 17-27. DOI: 10.1016/j.jacc.2020.05.016.
[79] Mehta JL, Bursac Z, Hauer-Jensen M, et al. Comparison of mortality rates in statin users versus nonstatin users in a United States veteran population[J]. Am J Cardiol, 2006, 98(7): 923-928.DOI: 10.1016/j.amjcard.2006.04.033.
[80] Savarese G, Gotto AJ, Paolillo S, et al. Benefits of statins in elderly subjects without established cardiovascular disease: a meta-analysis[J]. J Am Coll Cardiol, 2013, 62(22): 2090-2099.DOI: 10.1016/j.jacc.2013.07.069.
[81] Teng M, Lin L, Zhao YJ, et al. Statins for primary prevention of cardiovascular disease in elderly patients: systematic review and meta-analysis[J]. Drugs Aging, 2015, 32(8): 649-661.DOI: 10.1007/s40266-015-0290-9.
[82] Ridker PM, Lonn E, Paynter NP, et al. Primary prevention with statin therapy in the elderly: new meta-analyses from the contemporary JUPITER and HOPE-3 randomized trials[J]. Circulation, 2017, 135(20):1979-1981.DOI: 10.1161/CIRCULATIONAHA.117.028271.
[83] Miettinen TA, Pyorala K, Olsson AG, et al. Cholesterol-lowering therapy in women and elderly patients with myocardial infarction or angina pectoris: findings from the Scandinavian simvastatin survival study (4S)[J]. Circulation, 1997, 96(12):4211-4218.DOI: 10.1161/01.cir.96.12.4211.
[84] Lewis SJ, Moye LA, Sacks FM, et al. Effect of pravastatin on cardiovascular events in older patients with myocardial infarction and cholesterol levels in the average range. Results of the cholesterol and recurrent events (CARE) trial[J]. Ann Intern Med, 1998, 129(9):681-689.DOI: 10.7326/0003-4819-129-9-199811010-00002.
[85] Hunt D, Young P, Simes J, et al. Benefits of pravastatin on cardiovascular events and mortality in older patients with coronary heart disease are equal to or exceed those seen in younger patients: results from the LIPID trial[J]. Ann Intern Med, 2001, 134(10):931-940. DOI: 10.7326/0003-4819-134-10-200105150-00007.
[86] Heart Protection Study Collaborative Group. The effects of cholesterol lowering with simvastatin on cause-specific mortality and on cancer incidence in 20, 536 high-risk people: a randomised placebo-controlled trial [ISRCTN48489393][J]. BMC Med, 2005, 3:6. DOI: 10.1186/1741-7015-3-6
[87] Cannon CP, Braunwald E, Mccabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes[J]. N Engl J Med, 2004, 350(15):1495-1504. DOI: 10.1056/NEJMoa040583.
[88] Ray KK, Bach RG, Cannon CP, et al. Benefits of achieving the NCEP optional LDL-C goal among elderly patients with ACS[J]. Eur Heart J, 2006, 27(19):2310-2316.DOI: 10.1093/eurheartj/ehl180.
[89] Wenger NK, Lewis SJ, Herrington DM, et al. Outcomes of using high-or low-dose atorvastatin in patients 65 years of age or older with stable coronary heart disease[J]. Ann Intern Med, 2007, 147(1):1-9. DOI: 10.7326/147-1-200707030-00002.
[90] Ye P, Lu ZL, Du BM, et al. Effect of xuezhikang on cardiovascular events and mortality in elderly patients with a history of myocardial infarction: a subgroup analysis of elderly subjects from the China coronary secondary prevention study[J]. J Am Geriatr Soc, 2007, 55(7):1015-1022.DOI: 10.1111/j.1532-5415.2007.01230.x.
[91] Deedwania P, Stone PH, Bairey MC, et al. Effects of intensive versus moderate lipid-lowering therapy on myocardial ischemia in older patients with coronary heart disease: results of the study assessing goals in the elderly (SAGE)[J]. Circulation, 2007, 115(6): 700-707.DOI: 10.1161/CIRCULATIONAHA.106.654756.
[92] Olsson AG, Schwartz GG, Szarek M, et al. Effects of high-dose atorvastatin in patients>or=65 years of age with acute coronary syndrome (from the myocardial ischemia reduction with aggressive cholesterol lowering [MIRACL] study)[J]. Am J Cardiol, 2007, 99(5):632-635.DOI: 10.1016/j.amjcard.2006.09.111.
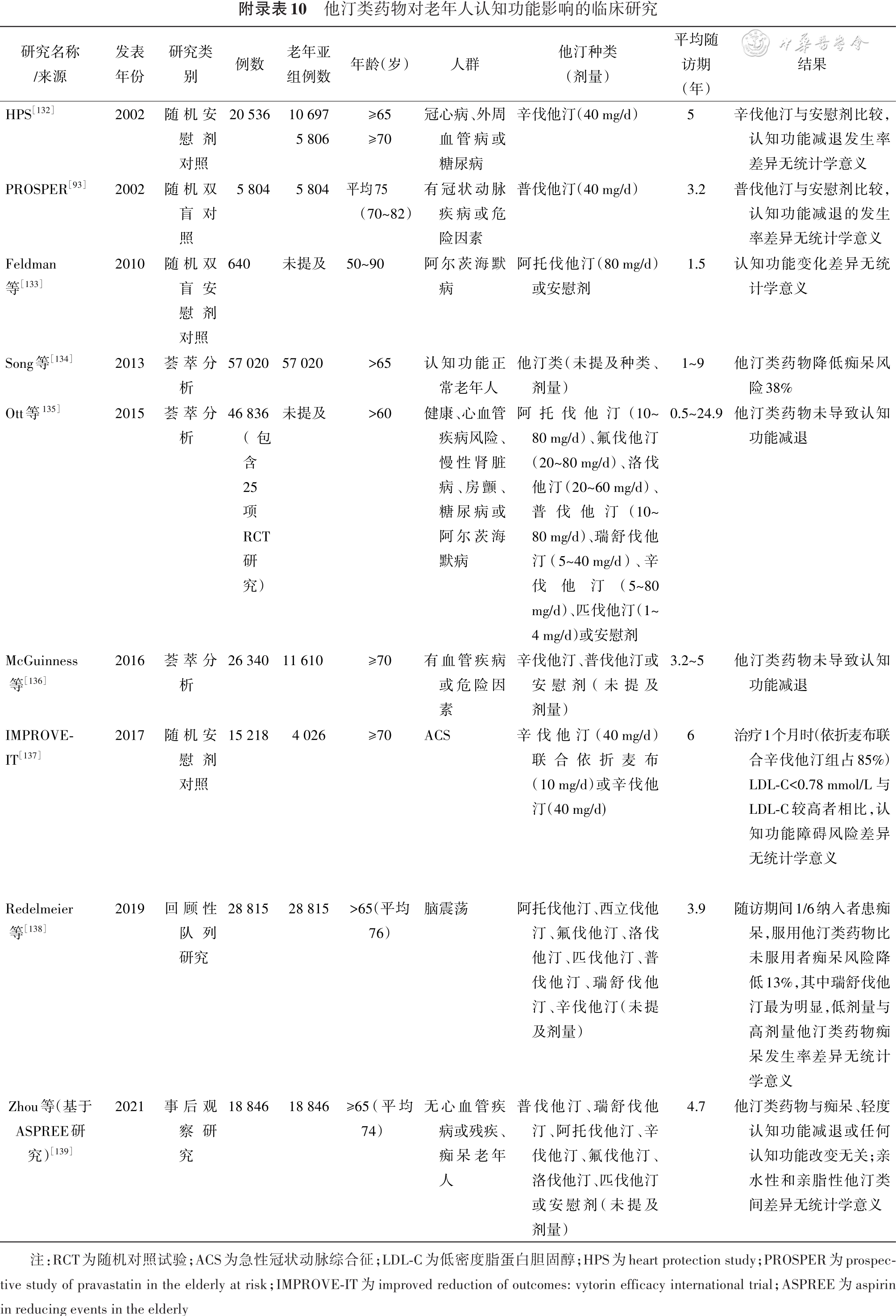
[93] Shepherd J, Blauw G J, Murphy M B, et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial[J]. Lancet, 2002, 360(9346):1623-1630. DOI: 10.1016/s0140-6736(02)11600-x.
[94] Lloyd SM, Stott DJ, de Craen AJ, et al. Long-term effects of statin treatment in elderly people: extended follow-up of the prospective study of pravastatin in the elderly at risk (PROSPER)[J]. PLoS One, 2013, 8(9):e72642.DOI: 10.1371/journal.pone.0072642.
[95] Eaton CB, Lapane KL, Murphy JB, et al. Effect of statin (HMG-Co-A-reductase inhibitor) use on 1-year mortality and hospitalization rates in older patients with cardiovascular disease living in nursing homes[J]. J Am Geriatr Soc, 2002, 50(8):1389-1395.DOI: 10.1046/j.1532-5415.2002.50360.x.
[96] Allen MC, Muhlestein JB, Horne BD, et al. Statin therapy is associated with reduced mortality across all age groups of individuals with significant coronary disease, including very elderly patients[J]. J Am Coll Cardiol, 2002, 40(10):1777-1785. DOI: 10.1016/s0735-1097(02)02477-4.
[97] Cooke CA, Kirkland SA, Sketris IS, et al. The impact of statins on health services utilization and mortality in older adults discharged from hospital with ischemic heart disease: a cohort study[J]. BMC Health Serv Res, 2009, 9:198. DOI: 10.1186/1472-6963-9-198.
[98] Gransbo K, Melander O, Wallentin L, et al. Cardiovascular and cancer mortality in very elderly post-myocardial infarction patients receiving statin treatment[J]. J Am Coll Cardiol, 2010, 55(13):1362-1369.DOI: 10.1016/j.jacc.2010.01.013.
[99] Afilalo J, Duque G, Steele R, et al. Statins for secondary prevention in elderly patients: a hierarchical bayesian meta-analysis[J]. J Am Coll Cardiol, 2008, 51(1):37-45.DOI: 10.1016/j.jacc.2007.06.063.
[100] Yan YL, Qiu B, Hu LJ, et al. Efficacy and safety evaluation of intensive statin therapy in older patients with coronary heart disease: a systematic review and meta-analysis[J]. Eur J Clin Pharmacol, 2013, 69(12):2001-2009.DOI: 10.1007/s00228-013-1570-0.
[101] Mizuno K, Nakaya N, Ohashi Y, et al. Usefulness of pravastatin in primary prevention of cardiovascular events in women: analysis of the Management of Elevated Cholesterol in the Primary Prevention Group of Adult Japanese (MEGA study)[J]. Circulation, 2008, 117(4):494-502. DOI: 10.1161/CIRCULATIONAHA.106.671826.
[102] Fulcher J, O′Connell R, Voysey M, et al. Efficacy and safety of LDL-lowering therapy among men and women: meta-analysis of individual data from 174, 000 participants in 27 randomised trials[J]. Lancet, 2015, 385(9976):1397-1405.DOI: 10.1016/S0140-6736(14)61368-4.
[103] Collins R, Armitage J, Parish S, et al. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebo-controlled trial[J]. Lancet, 2003, 361(9374):2005-2016. DOI: 10.1016/s0140-6736(03)13636-7.
[104] Neil HA, Demicco DA, Luo D, et al. Analysis of efficacy and safety in patients aged 65-75 years at randomization: collaborative atorvastatin diabetes study (CARDS)[J]. Diabetes Care, 2006, 29(11):2378-2384.DOI: 10.2337/dc06-0872.
[105] Kearney PM, Blackwell L, Collins R, et al. Efficacy of cholesterol-lowering therapy in 18, 686 people with diabetes in 14 randomised trials of statins: a meta-analysis[J]. Lancet, 2008, 371(9607):117-125.DOI: 10.1016/S0140-6736(08)60104-X.
[106] Wanner C, Krane V, Marz W, et al. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis[J]. N Engl J Med, 2005, 353(3):238-248. DOI: 10.1056/NEJMoa043545.
[107] Fellstrom BC, Jardine AG, Schmieder RE, et al. Rosuvastatin and cardiovascular events in patients undergoing hemodialysis[J]. N Engl J Med, 2009, 360(14):1395-1407. DOI: 10.1056/NEJMoa0810177.
[108] Baigent C, Landray MJ, Reith C, et al. The effects of lowering LDL cholesterol with simvastatin plus ezetimibe in patients with chronic kidney disease (study of heart and renal protection): a randomised placebo-controlled trial[J]. Lancet, 2011, 377(9784):2181-2192. DOI: 10.1016/S0140-6736(11)60739-3.
[109] Herrington W, Emberson J, Staplin N, et al. The effect of lowering LDL cholesterol on vascular access patency: post hoc analysis of the study of heart and renal protection[J]. Clin J Am Soc Nephrol, 2014, 9(5):914-919.DOI: 10.2215/CJN.10371013.
[110] Hou W, Lv J, Perkovic V, et al. Effect of statin therapy on cardiovascular and renal outcomes in patients with chronic kidney disease: a systematic review and meta-analysis[J]. Eur Heart J, 2013, 34(24):1807-1817.DOI: 10.1093/eurheartj/eht065.
[111] Herrington WG, Emberson J, Mihaylova B, et al. Impact of renal function on the effects of LDL cholesterol lowering with statin-based regimens: a meta-analysis of individual participant data from 28 randomised trials[J]. Lancet Diabetes Endocrinol, 2016, 4(10): 829-839. DOI: 10.1016/S2213-8587(16)30156-5.
[112] Hwang SD, Kim K, Kim YJ, et al. Effect of statins on cardiovascular complications in chronic kidney disease patients: a network meta-analysis[J]. Medicine (Baltimore), 2020, 99(22):e20061. DOI: 10.1097/MD.0000000000020061.
[113] Ouchi Y, Sasaki J, Arai H, et al. Ezetimibe lipid-lowering trial on prevention of atherosclerotic cardiovascular disease in 75 or older (EWTOPIA 75): a randomized, controlled trial[J]. Circulation, 2019, 140(12):992-1003. DOI: 10.1161/CIRCULATIONAHA.118.039415.
[114] Keech A, Simes RJ, Barter P, et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial[J]. Lancet, 2005, 366(9500):1849-1861. DOI: 10.1016/S0140-6736(05)67667-2.
[115] Elam MB, Ginsberg HN, Lovato LC, et al. Association of fenofibrate therapy with long-term cardiovascular risk in statin-treated patients with type 2 diabetes[J]. JAMA Cardiol, 2017, 2(4):370-380.DOI: 10.1001/jamacardio.2016.4828.
[116] Alperovitch A, Kurth T, Bertrand M, et al. Primary prevention with lipid lowering drugs and long term risk of vascular events in older people: population based cohort study[J]. BMJ, 2015, 350: h2335. DOI: 10.1136/bmj.h2335.
[117] Marchioli R, Barzi F, Bomba E, et al. Early protection against sudden death by n-3 polyunsaturated fatty acids after myocardial infarction: time-course analysis of the results of the Gruppo Italiano per lo Studio della Sopravvivenza nell′Infarto Miocardico (GISSI)-Prevenzione[J]. Circulation, 2002, 105(16):1897-1903. DOI: 10.1161/01.cir.0000014682.14181.f2.
[118] Yokoyama M, Origasa H, Matsuzaki M, et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): a randomised open-label, blinded endpoint analysis[J]. Lancet, 2007, 369(9567):1090-1098.DOI: 10.1016/S0140-6736(07)60527-3.
[119] Bowman L, Mafham M, Wallendszus K, et al. Effects of n-3 fatty acid supplements in diabetes mellitus[J]. N Engl J Med, 2018, 379(16):1540-1550. DOI: 10.1056/NEJMoa1804989.
[120] Manson JE, Cook NR, Lee IM, et al. Marine n-3 fatty acids and prevention of cardiovascular disease and cancer[J]. N Engl J Med, 2019, 380(1):23-32. DOI: 10.1056/NEJMoa1811403.
[121] Hu Y, Hu F B, Manson J E. Marine omega-3 supplementation and cardiovascular disease: an updated meta-analysis of 13 randomized controlled trials involving 127 477 participants[J]. J Am Heart Assoc, 2019, 8(19): e13543. DOI: 10.1161/JAHA.119.013543.
[122] Bernasconi AA, Wiest MM, Lavie CJ, et al. Effect of omega-3 dosage on cardiovascular outcomes: an updated meta-analysis and meta-regression of interventional trials[J]. Mayo Clin Proc, 2021, 96(2):304-313. DOI: 10.1016/j.mayocp.2020.08.034.
[123] Sattar N, Preiss D, Murray HM, et al. Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials[J]. Lancet, 2010, 375(9716):735-742. DOI: 10.1016/S0140-6736(09)61965-6.
[124] Ridker PM, Pradhan A, Macfadyen JG, et al. Cardiovascular benefits and diabetes risks of statin therapy in primary prevention: an analysis from the JUPITER trial[J]. Lancet, 2012, 380(9841): 565-571. DOI: 10.1016/S0140-6736(12)61190-8.
[125] Culver AL, Ockene IS, Balasubramanian R, et al. Statin use and risk of diabetes mellitus in postmenopausal women in the women′s health initiative[J]. Arch Intern Med, 2012, 172(2):144-152.DOI: 10.1001/archinternmed.2011.625.
[126] Wang KL, Liu CJ, Chao TF, et al. Statins, risk of diabetes, and implications on outcomes in the general population[J]. J Am Coll Cardiol, 2012, 60(14):1231-1238.DOI: 10.1016/j.jacc.2012.05.019.
[127] Zaharan NL, Williams D, Bennett K. Statins and risk of treated incident diabetes in a primary care population[J]. Br J Clin Pharmacol, 2013, 75(4):1118-1124.DOI: 10.1111/j.1365-2125.2012.04403.x.
[128] Carter AA, Gomes T, Camacho X, et al. Risk of incident diabetes among patients treated with statins: population based study[J]. BMJ, 2013, 346:f2610.DOI: 10.1136/bmj.f2610.
[129] Macedo AF, Douglas I, Smeeth L, et al. Statins and the risk of type 2 diabetes mellitus: cohort study using the UK clinical practice pesearch datalink[J]. BMC Cardiovasc Disord, 2014, 14: 85. DOI: 10.1186/1471-2261-14-85.
[130] Lin ZF, Wang CY, Shen LJ, et al. Statin use and the risk for incident diabetes mellitus in patients with acute coronary syndrome after percutaneous coronary intervention: a population-based retrospective cohort study in Taiwan[J]. Can J Diabetes, 2016, 40(3):264-269.DOI: 10.1016/j.jcjd.2015.12.006.
[131] Chen TL, Lin CS, Lin JA, et al. Evaluating risk of incident diabetes between patients who used lovastatin and red yeast rice prescriptions (lipocol forte): a retrospective cohort study based on a real-world database[J]. Diabetes Metab Syndr Obes, 2020, 13:89-98.DOI: 10.2147/DMSO.S223833.
[132] Heart Protection Study Collaborative Group. MRC/BHF heart protection study of cholesterol lowering with simvastatin in 20, 536 high-risk individuals: a randomised placebo-controlled trial[J]. Lancet, 2002, 360(9326): 7-22. DOI: 10.1016/S0140-6736(02)09327-3.
[133] Feldman HH, Doody RS, Kivipelto M, et al. Randomized controlled trial of atorvastatin in mild to moderate Alzheimer disease: LEADe[J]. Neurology, 2010, 74(12):956-964. DOI: 10.1212/WNL.0b013e3181d6476a.
[134] Song Y, Nie H, Xu Y, et al. Association of statin use with risk of dementia: a meta-analysis of prospective cohort studies[J]. Geriatr Gerontol Int, 2013, 13(4):817-824. DOI: 10.1111/ggi.12044.
[135] Ott BR, Daiello LA, Dahabreh IJ, et al. Do statins impair cognition? A systematic review and meta-analysis of randomized controlled trials[J]. J Gen Intern Med, 2015, 30(3):348-358. DOI: 10.1007/s11606-014-3115-3.
[136] Mcguinness B, Craig D, Bullock R, et al. Statins for the prevention of dementia[J]. Cochrane Database Syst Rev, 2016(1):D3160.DOI: 10.1002/14651858.CD003160.pub3.
[137] Giugliano RP, Wiviott SD, Blazing MA, et al. Long-term safety and efficacy of achieving very low levels of low-density lipoprotein cholesterol: a prespecified analysis of the IMPROVE-IT trial[J]. JAMA Cardiol, 2017, 2(5): 547-555. DOI: 10.1001/jamacardio.2017.0083.
[138] Redelmeier DA, Manzoor F, Thiruchelvam D. Association between statin use and risk of dementia after a concussion[J]. JAMA Neurol, 2019, 76(8):887-896.DOI: 10.1001/jamaneurol.2019.1148.
[139] Zhou Z, Ryan J, Ernst ME, et al. Effect of statin therapy on cognitive decline and incident dementia in older adults[J]. J Am Coll Cardiol, 2021, 77(25): 3145-3156. DOI: 10.1016/j.jacc.2021.04.075.
[140] Collins R, Armitage J, Parish S, et al. Effects of cholesterol-lowering with simvastatin on stroke and other major vascular events in 20536 people with cerebrovascular disease or other high-risk conditions[J]. Lancet, 2004, 363(9411):757-767.DOI: 10.1016/S0140-6736(04)15690-0.
[141] Amarenco P, Bogousslavsky J, Callahan AR, et al. High-dose atorvastatin after stroke or transient ischemic attack[J]. N Engl J Med, 2006, 355(6):549-559. DOI: 10.1056/NEJMoa061894.
[142] Goldstein LB, Amarenco P, Szarek M, et al. Hemorrhagic stroke in the stroke prevention by aggressive reduction in cholesterol levels study[J]. Neurology, 2008, 70(24 Pt 2):2364-2370. DOI: 10.1212/01.wnl.0000296277.63350.77.
[143] Amarenco P, Kim JS, Labreuche J, et al. Benefit of targeting a LDL (low-density lipoprotein) cholesterol<70 mg/dL during 5 years after ischemic stroke[J]. Stroke, 2020, 51(4):1231-1239. DOI: 10.1161/STROKEAHA.119.028718.
[144] Sanz-Cuesta BE, Saver JL. Lipid-lowering therapy and hemorrhagic stroke risk: comparative meta-analysis of statins and PCSK9 inhibitors[J]. Stroke. 2021, 52(10):3142-3150. DOI: 10.1161/STROKEAHA.121.034576.